 
Okay, so we know oxygen has 8 electrons, and only two electrons can exist in each orbital. This means thatĪnd if you were to go a step farther with helium, you would get 1s2, because it has two electrons in that s orbital. /complete-periodic-table-of-elements-royalty-free-vector-166052665-5a565f0e47c2660037ab8aca.jpg)
Hydrogen has an electron configuration of 1s1. Their electron configuration is determined by the number of electrons their neutral form has and the orbitals in which those electrons exist. The numbers that you are talking about are called electron configurations, and each element has its own unique one. And I really don't know much about f orbitals (I've never had one that large) but I believe there can be seven d orbitals are a really complicated shape and there can be up to five. p orbitals are kind of dumbbell shaped and there can be up to three of them at each energy level. s orbitals are sphere shaped, and there can only be one at each energy level. Okay, now, there are different types of orbitals that each take different shapes. Finally, each orbital can hold only two electrons and no more. The farther out the orbital from the nucleus, the more energy the electron has. 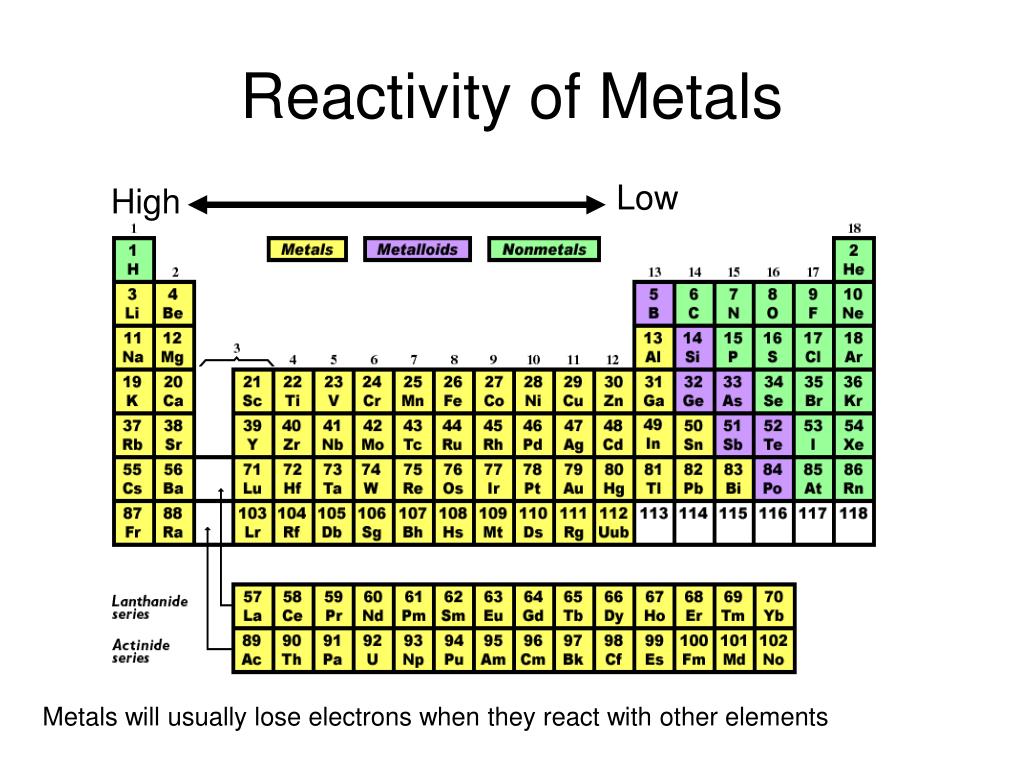 
Each orbital also represents a different energy level. For example, if the orbital is in the shape of a sphere, the electron can exist in any point within that sphere, but it cannot go outside that sphere. An orbital is basically a given space that an electron can exist in. You may already know some of this stuff but I'll go over all of it anyway.īasically, every atom has orbitals around their nucleus. In addition, these elements are colorless, odorless, and generally non-flammable.Okay, this is a long, long explanation. They are also considered to be chemically inert, meaning they are resistant to change. They are not easily oxidized and do not form compounds with other elements. These noble gases have full outer electron shells, which makes them highly unreactive. The least reactive elements on the periodic table are helium, neon, argon, krypton, xenon, and radon. Despite their differences, both noble gases and alkali metals are considered to be relatively nonreactive elements. The alkali metals, on the other hand, have only one valence electron, which makes them very reactive and prone to forming bonds. Noble gases are often considered the least reactive elements because they have full outer electron shells, which makes them resistant to bonding. The least reactive metals are found among the noble gases and the alkali metals at the far left of the periodic table. In this article, we'll explore the least reactive metals on the periodic table, how they differ from other metals, and how they can be used in everyday life. This means that the least reactive metals are found on the far left side of the table. Generally speaking, the farther left and down the periodic table one goes, the less reactive the elements become. Some metals are more reactive than others, meaning they will interact with other elements more readily than other metals. Reactivity refers to the tendency of an element to interact with other elements, molecules, and compounds. As such, knowing where to find the least reactive metals on the periodic table can be a great way to understand the chemistry of different elements. It is a useful resource for understanding the properties of different elements, including their reactivity. The periodic table is one of the most important tools in chemistry and materials science. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
