  
I would encourage you to explore further. 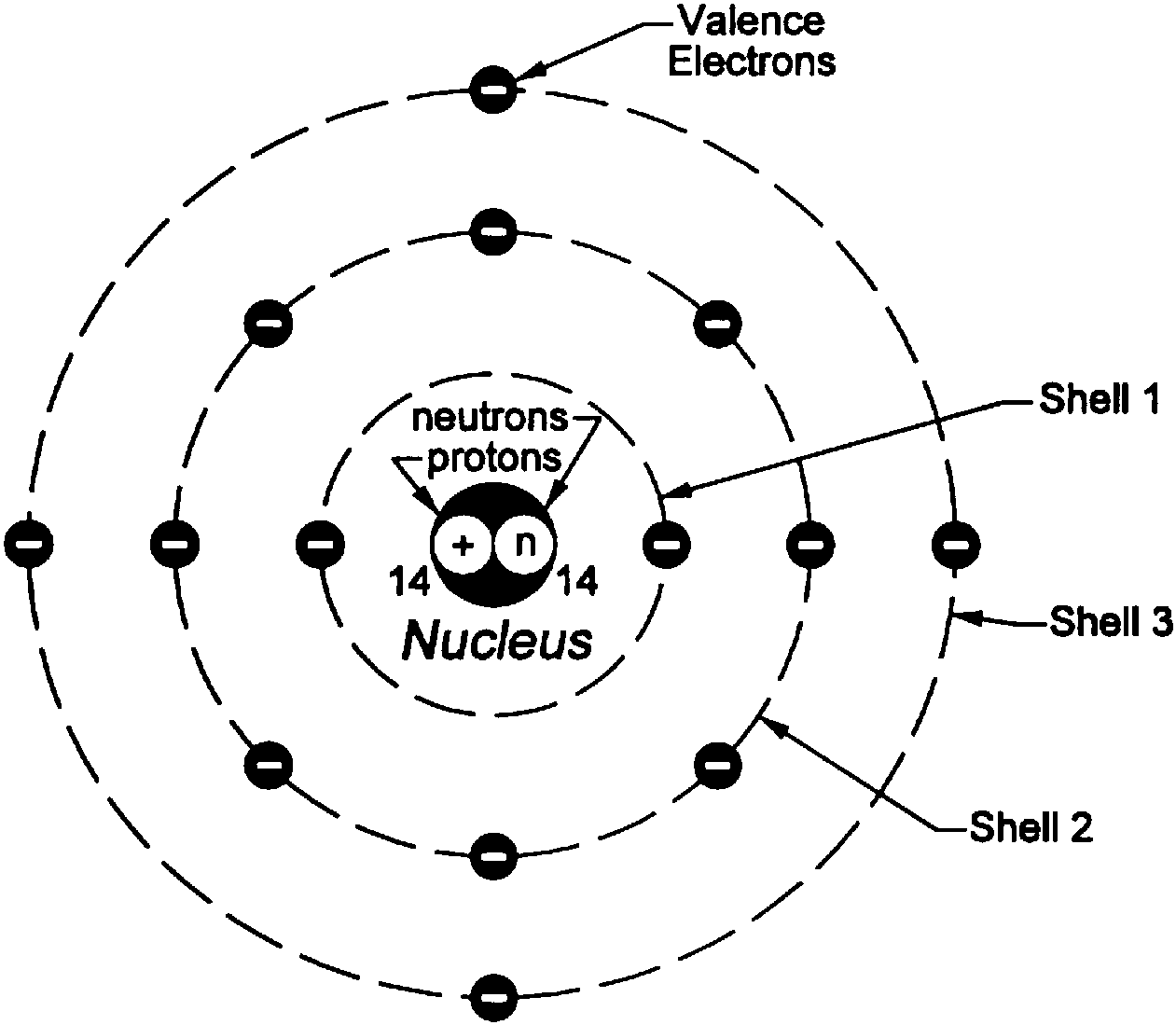
looking for relationships in a meaningful way. We can, however, say that the more electrons there are in an atom, the greater the variety of photon energies there will be so this may be a link worth exploringĪs I say, you are asking a great question. Es wurde 1915/16 von Arnold Sommerfeld vorgeschlagen und stellt eine Verfeinerung des Bohrschen Atommodells dar. Bohrs model of hydrogen is based on the nonclassical assumption that electrons travel in specific shells, or orbits, around the nucleus. as it switches from one energy level to another, then it emits / absorbs photons. Das Bohr-Sommerfeldsche Atommodell, Sommerfeldsche Atommodell oder die Sommerfeld-Erweiterung ist eine physikalische Beschreibung der Elektronenbahnen in einem Atom. This is because the electron can exist in many energy levels. Hydrogen, for example, although the simplest atom, has a whole range of photon energies that it emits. The value of the energies of photons does depend on the available energy levels in the atom. Now, the energy of the photon emitted from any element does not depend on the number of electrons in the atom. OK: I would say that the periodic table tells us about the number of protons in an element and, therefore the number of electrons too. Here is my answer, but I would encourage you to explore this and similar questions further. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
